An international team led by Jaime de Juan-Sanz at the Paris Brain Institute has shown that slightly increasing the metabolic capacity of neurons can enhance long-term memory in both fruit flies and mice. The study, published in Nature Metabolism, suggests that energy is not merely a prerequisite for brain function but may also serve as a lever to improve cognitive performance.
The brain is one of the most energy-demanding organs in the body. Whenever we articulate a thought, reason, or form a new memory, neurons become active. This activity requires fuel in the form of ATP, a molecule produced by mitochondria—the cell’s powerhouses. When a neuron is stimulated, intracellular calcium levels rise; part of this calcium enters the mitochondria, where it accelerates the Krebs cycle, a cascade of chemical reactions that boosts ATP production.
“The entry and exit of calcium within mitochondria allow energy production to be finely tuned to the demands of brain activity,” says Jaime de Juan-Sanz (CNRS), head of the PreSyn team at the Paris Brain Institute. “Until now, however, this mechanism had mainly been studied in situations that cause an energy deficit impairing information transmission at synapses.”
Working with colleagues from the Brain Plasticity Laboratory at ESPCI in Paris, the Hospital del Mar Research Institute in Barcelona, the Institute of Science and Technology in Vienna and the Max Planck Florida Institute for Neuroscience in Jupiter, the researchers asked a different question: what happens if we leverage mitochondrial calcium to increase energy production in neurons beyond what the brain needs to function normally?
Retaining Mitochondrial Calcium
To answer this question, the team focused on LETM1, a protein located in the inner mitochondrial membrane that helps export calcium from the mitochondrial matrix. By reducing its expression in cellular models, the researchers slowed calcium extrusion following neuronal activation. As a result, calcium remains longer inside the mitochondria, prolonging metabolic stimulation and leading to overproduction of ATP.
What makes this approach particularly interesting is that it extends a physiological cellular signal without overloading mitochondria with calcium, which could otherwise become toxic to neurons.
The team then examined the impact of this controlled metabolic boost in living animals. In both species studied, fruit flies and mice, LETM1 inhibition had a significant impact on behavior, particularly performance in tasks requiring long-term memory.
Memories That Last Longer
Normally, if a fruit fly experiences an odor paired with a mild punishment only once, it remembers the association for a few hours, but not the next day. To establish a lasting memory, the experiment must be repeated several times. However, researchers found that when LETM1 expression is reduced in mushroom body neurons (the center of olfactory memory in arthropods), a single training session is sufficient to produce a memory that persists for more than 24 hours.
“By contrast, we did not observe the same effect on middle-term memory. It appears that our manipulation does not enhance all forms of memory indiscriminately, but specifically those that require sustained energy investment,” the researcher adds.
The same type of Pavlovian conditioning was conducted in mice, with comparable results. This mechanism of memory consolidation appears to be conserved across very different species—especially given that the LETM1 protein is present in all eukaryotic organisms.
What If the Brain Could Work Better?
These findings suggest that slightly increasing the energy available to neurons can improve certain aspects of long-term memory performance. “Perhaps this phenomenon is not limited to memory and could also be used to stimulate other neural circuits—to improve cognitive endurance, for example,” argues Jaime de Juan-Sanz.
We are still far from being able to modulate the brain’s energy resources on demand. The genetic strategy used in this study is complex, and LETM1 dysregulation is implicated in several human diseases, including Wolf-Hirschhorn syndrome.
Nevertheless, the study invites a rethinking of energy’s role in brain function: it is not merely fuel, like a flashlight battery, but a genuine regulator of the intensity and duration of neuronal processes—more akin to a control panel.
“Most importantly, we have demonstrated that the brain may possess an unexpected capacity for optimization. That was far from obvious! The next step is to develop tools capable of modulating mitochondrial calcium with greater precision—for example, through optogenetics—to determine how far we can enhance memory consolidation,” concludes the researcher.
Sources
Vishwanath, A., et al. Mitochondrial Ca²⁺ efflux controls neuronal metabolism and long-term memory across species. Nature Metabolism, February 2026. DOI: 10.1038/s42255-026-01451-w.
Funding
This work was funded by the Diane Barrière Chair, the ERC Starting Grant, the ATIP-Avenir program, the Big Brain Theory program of the Paris Brain Institute, and the Kavli Exploratory Award.
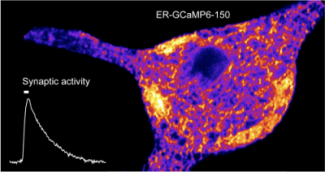
The team “Molecular physiology of presynaptic function” is studying the mitochondrial dysfunctions that may be responsible for epilepsy.
Read more