Why does the same genetic mutation cause a severe brain malformation in some patients but not in others? Researchers from the MOSAIC team at the Paris Brain Institute have developed mosaic human cortical organoids carrying mutations in the DEPDC5 gene in order to model focal cortical dysplasia—a brain malformation responsible for drug-resistant epilepsy in children. In a new study published in Brain, they show that biallelic inactivation of this gene is required to recapitulate the features of focal cortical dysplasia, and identify the key mechanisms at play.
Around 700,000 people in France live with epilepsy. Nearly half are under 20, and for one in three, available medications offer no relief. In some cases, the seizures are caused by a structural anomaly in the brain: focal cortical dysplasia type II (FCDII), a malformation that disrupts the organization of nerve cells in the cortex. For these patients, surgery-the removal of the seizure-generating area-is often the only option, with variable outcomes and significant risks.
FCDII belongs to a group of conditions known as ‘mTORopathies’, driven by mutations in genes that regulate cell growth and differentiation through the mTOR signaling pathway. These mutations can affect various cell types in the developing brain-neurons, astrocytes, or oligodendrocytes—producing a mosaic pattern: some cells carry the mutation, others do not.
A novel experimental model: mosaic mini-brains
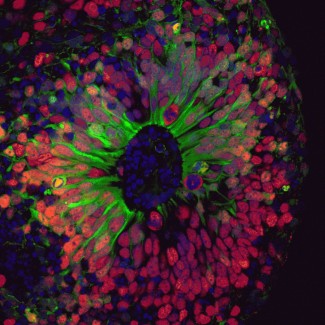
To uncover the mechanisms underlying FCDII, the Mosaic team at the Paris Brain Institute developed a new experimental approach. The researchers collected blood cells from a young patient carrying a mutation in the DEPDC5 gene-one of the main genes involved in FCDII-and from his unaffected brother, who served as a control. These cells were reprogrammed into induced pluripotent stem cells (iPSCs), then differentiated into human cortical organoids: tiny three-dimensional structures, a few millimeters across, that replicate key stages of human brain development.
“Mouse models of this condition existed, but they didn’t fully capture the complexity of the human patients. Human cortical development has features you simply don’t find in mice,” explains Marina Maletic, the study’s lead author, who worked on this project as part of her PhD. “So, we created human mosaic organoids by mixing cells carrying two mutated copies of the DEPDC5 with cells carrying only one. This allows us to recreate the genetic situation observed in patients’ brains.”
The team compared three types of organoids over a six-month development period: control (healthy) organoids, heterozygous organoids (with a single copy of the mutated gene), and the so-called mosaic organoids. This longitudinal comparison allowed the researchers to pinpoint which abnormalities require complete inactivation of the gene to emerge.
The case for a ‘two-hit’ mechanism
The findings confirm that a well-established theoretical framework in cancer genetics-the two-hit model, proposed by American geneticist Alfred G. Knudson in 1971-also applies to focal cortical dysplasia. The model holds that two successive ‘hits’ to DNA are needed to trigger the disease.
A single inherited mutation in the DEPDC5 gene is not sufficient to cause FCDII. It is the acquisition of a second spontaneous mutation-in a specific cell, at a specific moment in brain development-that sets off the pathological process.
Indeed, only the mosaic organoids reproduced the hallmarks of the condition: abnormally large neurons, accumulated neurofilaments, and hyperactivation of the mTOR pathway.
“Complete loss of DEPDC5 in a fraction of nerve cells is both necessary and sufficient to initiate the disease. The extent of the lesions then depends on the degree of mosaicism-in other words, on how many cells have sustained that second hit,” summarizes Marina Maletic.
Brain development disrupted from the earliest stages
To understand how the mutation disrupts brain development, the researchers examined the cellular state of their organoids at three key developmental time points-one month, three months, and six months-using a technique called single-cell RNA sequencing. This approach reveals which genes are active in each individual cell at any given moment, making it possible to detect disruptions that would otherwise be invisible.
The human cerebral cortex is built to a precise timetable: certain types of neurons must appear early, others later. The researchers found that this schedule was disrupted from the very first month, even in organoids carrying only a single mutated copy of the gene: neurons in the upper layers of the cortex appeared prematurely.
“This schedule was disrupted from the very first month, even in organoids carrying only a single mutated copy of the gene: neurons in the upper layers of the cortex appeared prematurely”, explains Sara Bizzotto, co-first author of the study.
Behind this acceleration: the loss of DEPDC5 abnormally activated genes that govern the pace of nerve cell maturation-in particular those involved in two major cell communication pathways, Notch and Wnt, which regulate the balance between stem cell proliferation and differentiation into mature neurons.
Measurable neuronal hyperactivity
Finally, spontaneous electrical activity was recorded from six-month organoids using a multi-electrode array. The mosaic organoids showed striking hyperactivity: their neurons fired more frequently and across a wider area than those in control or heterozygous organoids, suggesting pathological over-excitability of the neuronal network.
“You can’t really talk about epilepsy in an organoid. It’s just a model, not a real patient! But we do consider this abnormal electrical activity to be a correlate of epileptic seizures in humans. It rounds off our description of the pathological process,” adds Marina Maletic.
New avenues for research and treatment
Beyond their contribution to understanding FCDII, the researchers suggest that the dysregulated epilepsy-associated genes identified in this study may represent new therapeutic targets.
More broadly, mosaic organoids open the door to modeling other mosaic brain malformations whose study has long been hampered by the inaccessibility of human brain tissue.
“This is an excellent model that will ultimately enable precision medicine. By growing lab-based mini-brains from a patient’s own cells, we will be able to test multiple therapeutic options and identify which works best for that individual-without ever touching their brain,” she concludes.
Sources
Maletic, M., Bizzotto, S. et al. Mosaic human cortical organoids model 1 mTOR-related focal cortical dysplasia 2 through DEPDC5 deletion. Brain, Mars 2026. DOI : 10.1093/brain/awag086.
Funding
This work was supported by the European Research Council, the French ‘Investissements d’avenir’ program, the Fondation pour la Recherche Médicale (FRM), and the ERA-NET NEURON network.
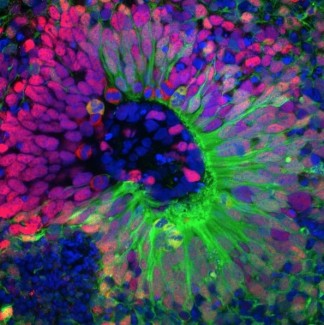
Image
Brain organoid. Credit: Marina Maletic.
The team "Genetic Mosaicism in Epilepsy and Neurodevelopmental Disorders" focuses on focal epilepsies and malformations of cortical development.
Read more